Decoding Goose MCP: Structure and Biological Role

In the intricate world of virology and molecular biology, the major capsid protein (MCP) stands as a cornerstone of viral architecture and function. These proteins are not merely structural components; they are critical players in the entire viral lifecycle, from host cell recognition and entry to genome packaging and immune evasion. When we speak of "Goose MCP," we delve into the specific context of viral infections affecting geese, a field of significant agricultural and ecological importance. Understanding the structure and biological role of these particular major capsid proteins offers profound insights into goose health, disease prevention, and potential therapeutic interventions. This extensive exploration will meticulously unravel the complexities of Goose MCP, examining its detailed structural characteristics, its multifaceted roles in viral pathogenesis, and the broader implications for virological research, while also briefly addressing other interpretations of the "MCP" acronym to ensure comprehensive coverage.
The study of viral major capsid proteins, particularly those found in avian species like geese, provides a window into host-pathogen interactions that are uniquely tailored to their ecological niches. Geese, being important poultry and waterfowl, are susceptible to a range of viral diseases, many of which are mediated by the major capsid proteins of the causative agents. These proteins are the primary interface between the virus and its host, dictating tropism, infectivity, and immunogenicity. Decoding their precise architecture and the intricate dance they perform within the host cell is paramount for developing effective diagnostic tools and robust prophylactic measures, such as vaccines. Without a deep understanding of the Goose MCP, our ability to combat diseases threatening goose populations, and by extension, the poultry industry, would be severely hampered. This article aims to provide a comprehensive, detailed examination of this crucial viral component, grounding the discussion in contemporary scientific understanding and exploring its far-reaching implications.
The Enigma of "Goose MCP": Initial Delimitation and Context
The acronym "MCP" can, in various contexts, refer to different entities. While this article's primary focus, as indicated by "Goose MCP" in the title, is unequivocally on the biological major capsid protein of viruses affecting geese, it is essential to acknowledge the existence of other interpretations for the "MCP" acronym. For instance, in the realm of computing and software, "MCP" could stand for "Model Context Protocol." This phrase refers to a defined set of rules and formats that govern how different computational models interact with their environment, data inputs, and other models within a larger system. Such protocols are crucial for ensuring interoperability, data consistency, and streamlined execution in complex software architectures, particularly those involving artificial intelligence and machine learning components. However, for the vast majority of this discourse, our attention remains firmly fixed on the biological entity.
Within the biological domain, "Goose MCP" predominantly refers to the major capsid protein of viruses that specifically infect geese. One of the most prominent examples where this concept is critically important is Goose Parvovirus (GPV). GPV is the etiological agent of Derzsy's disease, a highly contagious and often fatal disease affecting goslings and Muscovy ducklings. The parvovirus family, to which GPV belongs, typically encapsidates its single-stranded DNA genome within an icosahedral capsid composed of multiple copies of viral proteins (VPs), including major and minor capsid proteins. For GPV, the major capsid protein, often designated as VP2 or VP3 depending on the specific cleavage and maturation processes, is the most abundant protein forming the viral shell. This protein is responsible for the structural integrity of the virion and plays a pivotal role in mediating the initial interactions with host cells, making it a prime target for research into pathogenicity, diagnostics, and vaccine development. Therefore, our deep dive into "Goose MCP" will predominantly use GPV as a representative model, allowing for concrete examples and detailed discussions of its structure and biological activities.
The significance of understanding Goose MCP stems from several critical factors. Firstly, the poultry industry, including goose farming, contributes substantially to global food security and local economies. Diseases like Derzsy's disease can lead to catastrophic losses, severely impacting livelihoods and food supply chains. By unraveling the molecular mechanisms of GPV's Goose MCP, scientists can devise more effective strategies to control and prevent outbreaks. Secondly, the study of avian viruses, particularly those like GPV which can evolve rapidly, offers valuable insights into zoonotic potential and cross-species transmission, even if GPV itself is not known to be zoonotic. Understanding the host range determinants embedded within the MCP can inform broader efforts in pandemic preparedness. Finally, Goose MCP serves as an excellent model for studying fundamental principles of viral assembly, host receptor recognition, and immune evasion tactics employed by non-enveloped DNA viruses, contributing to general virological knowledge.
I. Fundamental Principles of Viral Major Capsid Proteins (MCPs): A Broader Perspective
Before delving into the specific characteristics of Goose MCP, it is crucial to establish a foundational understanding of major capsid proteins (MCPs) in general. These proteins are the primary building blocks of the viral capsid, the protective protein shell that encloses the viral genome. In non-enveloped viruses, the capsid is the outermost layer, directly interacting with the host environment. In enveloped viruses, the capsid is often encased within a lipid bilayer derived from the host cell membrane, though it still plays a crucial role in genome protection and sometimes in internal structural organization. The universal function of an MCP across all viral families is to protect the viral genetic material (DNA or RNA) from degradation by nucleases, extreme pH, temperature fluctuations, and mechanical shear forces outside the host cell. This protective role is absolutely essential for the survival and transmission of the virus.
Beyond mere protection, MCPs are highly versatile proteins that orchestrate a myriad of functions throughout the viral lifecycle. They are responsible for the precise packaging of the viral genome, a process that is often highly regulated and involves specific recognition sequences within the genome and corresponding binding domains on the MCP. This packaging ensures that each progeny virion receives a complete and functional copy of the genetic blueprint. Furthermore, MCPs are the primary determinants of viral tropism, dictating which host cells or tissues a virus can infect. This specificity arises from the ability of MCPs to recognize and bind to specific cell surface receptors on target cells. This initial binding event is the first critical step in infection, triggering subsequent events such as receptor-mediated endocytosis or direct membrane fusion for enveloped viruses, facilitating viral entry into the host cell. The exquisite specificity of these interactions is a key factor in viral pathogenesis and host range.
The diversity of MCPs across the viral kingdom is astounding, reflecting millions of years of evolutionary adaptation to different hosts and environments. Despite this vast diversity, certain fundamental principles of assembly and structural organization are conserved. Many viruses, particularly those with isometric (roughly spherical) capsids, adhere to icosahedral symmetry, a highly efficient way to construct a closed shell from multiple identical or quasi-identical protein subunits. This symmetry minimizes the amount of genetic information required to encode the capsid and maximizes stability. The number of MCP subunits can range from 12 in the smallest viruses (e.g., adeno-associated virus, AAV) to hundreds or even thousands in larger viruses (e.g., herpesviruses, adenoviruses). Each subunit, or a combination of subunits (capsomers), is precisely positioned to form the intricate viral lattice. Helical symmetry is another common architectural principle, particularly in RNA viruses, where MCPs assemble around the RNA genome in a spiral fashion, forming rod-like or filamentous particles. The specific symmetry, subunit arrangement, and overall size of the capsid are all dictated by the inherent structural properties of the individual MCPs.
The study of MCPs relies heavily on advanced structural biology techniques, which have revolutionized our understanding of viral architecture at atomic resolution. Cryo-electron microscopy (cryo-EM) has emerged as a particularly powerful tool, allowing for the visualization of intact virions and their components in near-native states. This technique can reveal the precise arrangement of MCPs, the presence of minor capsid proteins, and even the internal organization of the genome. X-ray crystallography, while often requiring protein crystallization (which can be challenging for large, complex proteins), provides even higher resolution structural data, revealing atomic details of protein folds, active sites, and ligand-binding pockets. Nuclear Magnetic Resonance (NMR) spectroscopy is useful for studying smaller, more dynamic MCP domains or isolated proteins in solution. Complementing these experimental approaches, bioinformatics and computational modeling play an increasingly vital role. Sequence analysis allows for the identification of conserved motifs, prediction of secondary structures, and phylogenetic relationships. Molecular dynamics simulations can model the flexibility and interactions of MCPs, providing insights into assembly pathways and conformational changes during infection. Together, these methodologies paint a comprehensive picture of MCP structure and function, laying the groundwork for understanding the specificities of Goose MCP.
II. Unveiling the Structure of Goose MCP
Understanding the structure of Goose MCP is fundamental to comprehending its various biological roles. Like other major capsid proteins, Goose MCP, particularly that of Goose Parvovirus (GPV), exhibits a hierarchical organization, from its primary amino acid sequence to its intricate quaternary arrangement within the virion. Each level of this structural hierarchy contributes critically to the protein's overall function and stability.
A. Primary Structure: The Blueprint of Functionality
The primary structure of Goose MCP refers to its linear sequence of amino acids. For GPV, the major capsid protein, often designated VP2 or VP3 (depending on post-translational processing), typically comprises hundreds of amino acid residues. This sequence is not random; it is the ultimate determinant of all higher-order structures and functions. Bioinformatics tools are crucial for analyzing this sequence. For instance, sequence alignments with other parvoviruses reveal highly conserved regions, which often correspond to crucial functional domains, such as those involved in genome binding, inter-subunit interactions, or receptor recognition. Conversely, hypervariable regions are often found on the surface loops of the capsid, which are exposed to the host immune system. These variable regions contribute to antigenic diversity, allowing viruses to evade immune surveillance or adapt to new hosts.
Analysis of the GPV MCP primary sequence often reveals distinctive patterns: stretches of hydrophobic residues tend to form the core of the protein, contributing to its stability within the aqueous environment, while hydrophilic residues are often found on the surface, facilitating interactions with water and other cellular components. Specific amino acid motifs can also indicate potential post-translational modification sites, such as serine, threonine, or tyrosine residues for phosphorylation, or asparagine residues for N-linked glycosylation. These modifications can significantly alter the protein's charge, conformation, and interaction profile, influencing assembly, stability, and antigenicity. The exact length and amino acid composition vary slightly among different GPV strains, reflecting ongoing evolutionary pressures and adaptations.
B. Secondary Structure: Local Folds and Motifs
The secondary structure refers to the local folding patterns of the polypeptide chain, primarily alpha-helices and beta-sheets, stabilized by hydrogen bonds between backbone atoms. These elements are predicted from the primary sequence using computational algorithms and confirmed by structural determination methods. For many parvovirus MCPs, including GPV's, a characteristic beta-barrel motif (often referred to as an "eight-stranded anti-parallel beta-barrel" or "jelly-roll" motif) forms the core structural domain of each subunit. This motif is highly conserved across diverse viral families and provides exceptional stability to the capsid.
In addition to the beta-barrel, Goose MCP will also feature various loops connecting these secondary structure elements, as well as shorter alpha-helices. These loops, particularly those on the exterior of the protein, are often highly flexible and are critical for functional interactions. They can form the binding sites for host cell receptors, dictate antigenic epitopes recognized by antibodies, or participate in the intricate assembly process. The precise arrangement and connectivity of these secondary structures dictate the overall shape and rigidity of the individual MCP subunit, which is a prerequisite for its correct assembly into the larger capsid. Deviations in these local folds can lead to misfolding, aggregation, or loss of function, highlighting the critical importance of a correctly formed secondary structure.
C. Tertiary Structure: The Three-Dimensional Blueprint
The tertiary structure describes the overall three-dimensional folding of a single Goose MCP polypeptide chain, including the arrangement of all secondary structure elements and their interconnecting loops in space. This complex 3D shape is stabilized by a combination of forces, including hydrophobic interactions, hydrogen bonds, disulfide bridges (between cysteine residues), and ionic bonds. The tertiary structure dictates the presence and conformation of specific functional domains. For GPV MCP, these domains might include:
- Receptor-binding domains: Exposed loops or surface protrusions that specifically recognize and bind to host cell surface molecules. The precise shape and charge distribution of these domains are critical for host tropism.
- DNA-binding domains: Internal regions that interact with the viral genome, facilitating its packaging. These often contain positively charged residues to neutralize the negative charge of the nucleic acid backbone.
- Inter-subunit interaction domains: Surfaces or interfaces that mediate precise contacts with other MCP subunits during capsid assembly. These interactions are highly specific and crucial for forming the correct symmetrical structure.
The folding of Goose MCP into its functional tertiary structure is a highly orchestrated process, often involving cellular chaperones to ensure correct conformation. Mis-folded proteins can lead to non-infectious virions or trigger cellular stress responses. High-resolution structural techniques like cryo-EM and X-ray crystallography are essential for mapping these tertiary structures, revealing the atomic details of these critical functional domains and providing targets for antiviral drug design.
D. Quaternary Structure: The Assembled Virion
The quaternary structure refers to the arrangement of multiple Goose MCP subunits to form the complete viral capsid. For GPV, as a parvovirus, the capsid is typically an icosahedron, a polyhedron with 20 triangular faces and 12 vertices, exhibiting 5-3-2 rotational symmetry. This highly stable and efficient architecture is achieved by the precise assembly of 60 individual MCP subunits. In some parvoviruses, three MCP proteins (VP1, VP2, VP3) are derived from the same gene through alternative splicing and proteolytic cleavage. VP3 is typically the most abundant major capsid protein. These subunits come together to form pentamers (groups of five proteins) which then arrange themselves to form the icosahedral shell.
The assembly process is intricate, often beginning in the host cell nucleus (for DNA viruses like GPV) where the individual MCPs are synthesized and then transported. These proteins self-assemble in a highly coordinated fashion, driven by specific protein-protein interactions between adjacent subunits. The precise interfaces between the MCPs determine the overall stability and integrity of the capsid. During or after assembly, the viral genome is typically packaged into the pre-formed capsid, often through a dedicated portal or channel. The fidelity of this assembly process is paramount for producing infectious virions; even minor errors can lead to non-functional or unstable particles, highlighting the critical role of each Goose MCP subunit in the overall viral architecture.
E. Post-Translational Modifications: Fine-Tuning Function
Post-translational modifications (PTMs) are chemical alterations to the amino acid side chains of Goose MCP after its synthesis. These modifications can dramatically alter the protein's conformation, stability, localization, and interactions, effectively fine-tuning its biological function. Common PTMs include:
- Phosphorylation: The addition of a phosphate group, typically to serine, threonine, or tyrosine residues. Phosphorylation can act as a molecular switch, altering protein activity, driving conformational changes, or regulating protein-protein interactions critical for assembly or uncoating. For instance, phosphorylation of specific residues on parvovirus MCPs can influence nuclear import or interaction with host factors.
- Glycosylation: The addition of sugar moieties. While less common for internal viral capsid proteins of non-enveloped viruses, surface-exposed loops could potentially be glycosylated, especially if the protein interacts with the endoplasmic reticulum/Golgi apparatus. Glycosylation can affect antigenicity and receptor binding.
- Proteolytic Cleavage: The enzymatic removal of a portion of the polypeptide chain. For many parvoviruses, including GPV, a larger precursor protein (e.g., VP0) is cleaved by viral or host proteases to yield the mature capsid proteins (e.g., VP2 and VP3). This cleavage is often a maturation step, making the virion infectious by exposing functional domains or facilitating conformational changes necessary for uncoating.
- Acetylation, Ubiquitination: While less extensively studied for Goose MCP specifically, these modifications are common regulatory mechanisms for many cellular and viral proteins, influencing stability, trafficking, and interaction with cellular machinery.
The interplay of these PTMs determines the precise functional state of Goose MCP at different stages of the viral lifecycle, representing an additional layer of complexity in understanding its biological roles.
Table: Comparative Structural Features of Goose Parvovirus (GPV) MCP and a Model Mammalian Parvovirus (e.g., Canine Parvovirus, CPV) VP2
| Feature | Goose Parvovirus (GPV) MCP (e.g., VP3) | Canine Parvovirus (CPV) VP2 (Model Mammalian Parvovirus) |
|---|---|---|
| Viral Family | Parvoviridae, subfamily Parvovirinae, genus Anseriparvovirus | Parvoviridae, subfamily Parvovirinae, genus Protoparvovirus |
| Genome Type | Single-stranded DNA (ssDNA) | Single-stranded DNA (ssDNA) |
| Capsid Symmetry | Icosahedral (T=1, non-enveloped) | Icosahedral (T=1, non-enveloped) |
| Primary Structure | ~550-600 amino acids; strain-specific variations in hypervariable loops. | ~580 amino acids; distinct antigenic variations among strains (e.g., CPV-2a/b/c). |
| Core Secondary Struct. | Conserved eight-stranded anti-parallel beta-barrel ("jelly-roll" motif). | Conserved eight-stranded anti-parallel beta-barrel ("jelly-roll" motif). |
| Surface Loops | Highly variable; determine host specificity (goose/duck) and antigenicity. | Highly variable; determine host specificity (dog, cat) and major antigenic sites. |
| Receptor Binding | Specific interactions with goose/duck cell surface receptors (e.g., transferrin receptor-like molecule). | Specific interactions with canine/feline transferrin receptor 1 (TfR1). |
| Genome Packaging | Internal positive charges and specific motifs facilitate ssDNA packaging. | Internal positive charges facilitate ssDNA packaging. |
| Assembly | Self-assembles into 60 subunits (often VP3) to form the capsid in the nucleus. | Self-assembles into 60 subunits (VP2 and minor VP1) to form the capsid in the nucleus. |
| Post-Translational Mod. | Proteolytic cleavage from VP0 precursor; potential phosphorylation. | Proteolytic cleavage from VP0 precursor to VP2/VP3; phosphorylation is known to occur. |
| Antigenicity | Major target for goose immune response; forms basis for diagnostics and vaccines. | Major target for canine/feline immune response; forms basis for diagnostics and vaccines. |
III. The Multifaceted Biological Role of Goose MCP
The Goose MCP, particularly that of GPV, is far more than a simple structural component. It is a highly evolved protein that plays crucial roles at every stage of the viral lifecycle, orchestrating the complex interactions required for successful infection, replication, and transmission. Its functions range from the protection of the viral genome to the evasion of host immune responses, each contributing to the virus's pathogenicity and survival.
A. Genome Packaging and Protection: The Core Mission
The most fundamental role of Goose MCP is the encapsulation and protection of the viral genome. For GPV, this involves precisely packaging a single-stranded DNA molecule within the robust icosahedral capsid. This process is highly specific and efficient. The interior surfaces of the Goose MCP subunits often contain positively charged amino acid residues (like lysine and arginine) that can electrostatically interact with the negatively charged phosphate backbone of the DNA. These interactions are crucial not only for neutralizing the charge and compacting the genome but also for providing stability to the nucleocapsid complex.
The packaging process itself is an intricate dance. It often involves a packaging motor (not necessarily part of the MCP itself but interacting with it) that actively spools the genome into the pre-formed or assembling capsid. The MCP's role here is to provide the stable scaffold into which the genome is inserted and then to seal the entry pore once packaging is complete. The integrity of this protective shell is paramount; it shields the delicate DNA from nucleases present in the extracellular environment and from physical damage, ensuring that the viral genetic information reaches a new host cell intact and ready for replication. Any defect in the MCP's ability to form a stable capsid or package the genome effectively would render the virion non-infectious, highlighting the indispensable nature of this primary function.
B. Host Cell Attachment and Entry: Initiating Infection
The Goose MCP serves as the primary interface between the virus and its host cell, initiating the infection process through specific recognition and binding. For GPV, the surface-exposed loops and protrusions of its MCP are finely tuned to recognize and bind to specific receptor molecules on the surface of goose and Muscovy duckling cells. While the exact identity of the primary receptor for GPV is still under active investigation, studies on other parvoviruses suggest that common cellular components like transferrin receptor 1 (TfR1) or other cell surface glycoproteins or glycans can serve as entry receptors. The specificity of this binding interaction dictates the host range and tissue tropism of the virus.
Upon binding to the primary receptor, a series of conformational changes within the Goose MCP can be triggered, which are essential for subsequent entry events. This might involve additional binding to co-receptors or the internalization of the virion through endocytosis. For many non-enveloped viruses like GPV, clathrin-mediated endocytosis is a common pathway. The virion is taken up into an endosomal vesicle, and as the endosome acidifies, further conformational changes in the MCP can occur. These changes are crucial for exposing hydrophobic regions that facilitate the escape of the viral genome from the endosome into the cytoplasm, or in some cases, direct delivery to the nucleus. Without the precise execution of host cell attachment and entry, mediated solely by the Goose MCP, the virus cannot deliver its genetic payload and establish a productive infection.
C. Capsid Assembly and Maturation: Building the Virion
The assembly of hundreds of individual Goose MCP subunits into a symmetrical and stable capsid is one of the most remarkable feats of viral biology. This process is a highly ordered self-assembly pathway, often occurring within specific cellular compartments, such as the nucleus for GPV. Individual MCPs, after being synthesized in the cytoplasm and imported into the nucleus, begin to interact with each other. These interactions are highly specific, guided by the precise protein-protein interfaces encoded within the tertiary structure of each subunit.
The assembly often proceeds through intermediate structures, such as pentamers (five MCP subunits) or larger procapsids (empty capsid precursors). Cellular chaperones might assist in ensuring correct folding and preventing premature aggregation. Once the procapsid is formed, the viral genome is typically packaged, a step that might trigger further maturation events. For parvoviruses, this often involves the proteolytic cleavage of a precursor protein (e.g., VP0) into the mature MCPs (e.g., VP2 and VP3). This cleavage is not merely a cosmetic change; it often induces critical conformational shifts in the capsid that are necessary for its stability, receptor binding, or the subsequent uncoating process upon infection of a new cell. The maturation step ensures that the newly formed virions are fully infectious and capable of initiating another round of replication, underscoring the dynamic and crucial role of the Goose MCP in the complete viral lifecycle.
D. Immune Evasion and Modulation: Navigating Host Defenses
The Goose MCP, being the outermost component of the virion, is the primary target for the host's adaptive immune response, particularly neutralizing antibodies. However, viruses have evolved sophisticated mechanisms to evade or modulate these responses, and the Goose MCP plays a central role in these strategies. The hypervariable regions on the surface loops of the MCP, which are exposed to the immune system, can undergo rapid mutation. These mutations can alter the antigenic epitopes recognized by antibodies, allowing the virus to escape neutralization by pre-existing immunity. This antigenic drift is a common phenomenon observed in many viruses and can necessitate the periodic updating of vaccine strains.
Beyond structural variation, Goose MCP might also possess specific domains or motifs that interact with host immune components. For example, some viral proteins can bind to complement proteins, either activating or inhibiting the complement cascade, a critical part of the innate immune system. Others might interfere with antigen presentation pathways, preventing the host cell from effectively displaying viral peptides to T lymphocytes. While specific immune evasion mechanisms of GPV Goose MCP are still under detailed investigation, analogous strategies have been identified in other parvoviruses. Understanding how Goose MCP interacts with and potentially manipulates the goose immune system is vital for designing more effective vaccines that elicit broad and durable protective responses, or for developing immunomodulatory therapeutics.
E. Viral Replication Cycle Support: Beyond Structural Roles
While primarily a structural protein, Goose MCP can also contribute to other aspects of the viral replication cycle beyond capsid formation. For DNA viruses like GPV, the MCP might play a role in nuclear localization. After entering the cytoplasm, the capsid or partially uncoated genome needs to be transported into the nucleus, where DNA replication and transcription occur. Nuclear localization signals (NLS) present on the MCP can facilitate interaction with host nuclear import machinery, ensuring the efficient delivery of the viral genome to the site of replication.
Furthermore, some capsid proteins have been shown to interact directly with host cellular machinery or even viral non-structural proteins, influencing various steps of the replication process. For instance, they might regulate viral gene expression, enhance DNA replication efficiency, or participate in the assembly of replication factories. While these non-structural roles of Goose MCP in GPV are less characterized than its structural functions, the complexity of viral-host interactions suggests that MCPs are often multitasking proteins, contributing to the overall efficiency and success of the viral lifecycle in numerous subtle ways. Elucidating these secondary roles can uncover novel targets for antiviral intervention.
F. Pathogenesis: Contributing to Disease Development
The Goose MCP's structure and function are inextricably linked to the pathogenesis of diseases like Derzsy's disease caused by GPV. The ability of the MCP to bind specifically to target cells in geese (e.g., in the intestine, bursa of Fabricius, and other lymphoid organs) directly dictates the tropism and initial site of infection. Efficient entry into these cells, mediated by the MCP, allows the virus to establish a robust replication cycle, leading to cell lysis and tissue damage. The symptoms of Derzsy's disease, such as anorexia, depression, diarrhea, and ascites, are direct consequences of the widespread cellular destruction and inflammation initiated by viral replication, where the MCP is the primary agent of cellular invasion.
Moreover, the immunogenicity of the Goose MCP contributes to the host's pathological response. While a strong antibody response against the MCP is desired for protection, in some cases, immune responses can also contribute to tissue damage or immunopathology. The specific regions on the MCP that elicit neutralizing antibodies are of critical importance for vaccine design. By understanding which parts of the Goose MCP are essential for virulence and which elicit the most protective immune responses, researchers can rationally design attenuated live vaccines or subunit vaccines that effectively protect geese without causing adverse effects, thereby mitigating the severe impact of diseases like Derzsy's disease on poultry farming.
APIPark is a high-performance AI gateway that allows you to securely access the most comprehensive LLM APIs globally on the APIPark platform, including OpenAI, Anthropic, Mistral, Llama2, Google Gemini, and more.Try APIPark now! 👇👇👇
IV. Methodologies for Characterizing Goose MCP
The comprehensive understanding of Goose MCP's structure and function is a testament to the application of a diverse array of advanced scientific methodologies. These techniques span molecular biology, structural biology, biophysics, immunology, and computational sciences, each contributing unique insights into different facets of this complex viral protein.
A. Molecular Biology Techniques: Manipulating the Genetic Code
Molecular biology techniques form the bedrock of Goose MCP characterization, allowing scientists to manipulate its genetic blueprint. * Cloning and Expression: The gene encoding Goose MCP is typically cloned into expression vectors, allowing for its production in heterologous systems such as bacteria (e.g., E. coli), insect cells (e.g., baculovirus system), or mammalian cells. This enables the production of large quantities of purified MCP for biochemical and structural studies, free from other viral or host proteins. * Mutagenesis: Site-directed mutagenesis allows for precise changes to be made to specific amino acids within the Goose MCP sequence. By altering key residues suspected to be involved in receptor binding, DNA packaging, or inter-subunit interactions, researchers can dissect the functional importance of these amino acids. For instance, mutating a positively charged residue in a putative DNA-binding domain and observing a loss of packaging efficiency would confirm its role. * Recombinant Virus Generation: Advanced techniques allow for the generation of recombinant GPV or other goose-infecting viruses where the MCP gene has been modified. This can be used to study the impact of MCP mutations on viral infectivity, tropism, or pathogenesis in a native viral context. It also allows for the creation of attenuated vaccine candidates. * Reporter Assays: Fusing the Goose MCP gene or specific domains to reporter genes (e.g., GFP, luciferase) allows for studies on protein localization, expression levels, and interactions with host factors in live cells, providing dynamic insights.
B. Structural Biology: Visualizing at Atomic Resolution
Structural biology techniques are indispensable for revealing the three-dimensional architecture of Goose MCP. * Cryo-electron Microscopy (Cryo-EM): This technique has revolutionized viral structural biology. By flash-freezing virions or assembled capsids and imaging them with electron beams, cryo-EM can provide high-resolution 3D reconstructions of the entire Goose MCP-composed capsid, often to near-atomic resolution. This allows for the visualization of subunit arrangement, surface features (e.g., receptor-binding sites), and internal organization, including packaged genome. The advantage is that it does not require crystallization, making it suitable for large, complex biological assemblies. * X-ray Crystallography: If purified Goose MCP (or domains thereof) can be crystallized, X-ray diffraction patterns can be collected and processed to yield atomic-resolution structures. This technique provides exquisite detail about the backbone and side-chain conformations, disulfide bonds, and ligand-binding pockets, offering a deep understanding of protein folding and interactions. * Nuclear Magnetic Resonance (NMR) Spectroscopy: For smaller, soluble domains of Goose MCP, NMR can provide insights into solution-state structure and dynamics. It is particularly useful for studying flexible loops or protein-ligand interactions that might be difficult to resolve by crystallography.
C. Biophysical Approaches: Dynamics and Interactions
Biophysical methods quantify the physical properties and interactions of Goose MCP. * Analytical Ultracentrifugation (AUC): Measures the sedimentation rate and equilibrium distribution of proteins in a centrifugal field, providing information on molecular weight, shape, and oligomerization states (e.g., whether MCP exists as monomers, dimers, or pentamers in solution). * Dynamic Light Scattering (DLS): Used to determine the size distribution of particles in solution, helpful for monitoring the assembly of MCPs into capsids or detecting aggregation. * Surface Plasmon Resonance (SPR) or Bio-Layer Interferometry (BLI): These techniques measure real-time binding kinetics (association and dissociation rates) and affinities between Goose MCP and its potential binding partners, such as host cell receptors or neutralizing antibodies, providing quantitative data on molecular interactions. * Mass Spectrometry (MS): Used to confirm the molecular weight of purified Goose MCP, identify post-translational modifications (e.g., phosphorylation sites), and analyze protein complexes, providing detailed biochemical characterization.
D. Immunological Assays: Detecting and Responding
Immunological assays are crucial for understanding the host's response to Goose MCP and for diagnostic development. * Enzyme-Linked Immunosorbent Assay (ELISA): Uses purified Goose MCP as an antigen to detect antibodies against GPV in goose serum (for serodiagnosis) or to quantify the production of specific antibodies after vaccination. Conversely, antibodies against Goose MCP can be used to detect the presence of viral antigen in infected tissues. * Western Blot: Detects specific Goose MCP protein in complex mixtures (e.g., cell lysates, tissue samples) using antibodies, confirming its presence, size, and post-translational modifications. * Neutralization Assays: Measures the ability of antibodies (raised against Goose MCP or from infected animals) to neutralize the infectivity of GPV in cell culture. This is the gold standard for assessing the protective efficacy of vaccine-induced antibodies. * Immunofluorescence/Immunohistochemistry: Uses antibodies labeled with fluorescent dyes or enzymes to visualize the localization of Goose MCP within infected cells or tissues, providing insights into viral trafficking and pathology.
E. Computational Approaches: Prediction and Modeling
Computational methods are increasingly integrated into Goose MCP research, complementing experimental data. * Bioinformatics Sequence Analysis: Tools are used for sequence alignment, identification of conserved domains, prediction of secondary and tertiary structures, phylogenetic analysis (to understand evolutionary relationships between GPV strains), and identification of potential PTM sites. * Homology Modeling: If the structure of a closely related parvovirus MCP is known, homology modeling can be used to predict the 3D structure of Goose MCP based on sequence similarity, providing a starting point for further experimental validation. * Molecular Dynamics (MD) Simulations: These simulations model the movement and interactions of atoms and molecules over time, allowing researchers to study the flexibility of Goose MCP, conformational changes during receptor binding or uncoating, and the dynamics of capsid assembly. * Epitope Prediction: Algorithms can predict potential B-cell and T-cell epitopes on the Goose MCP, guiding the design of subunit vaccines or diagnostic antigens.
The synergistic application of these diverse methodologies provides a holistic view of Goose MCP, from its atomic architecture to its dynamic interactions within a living host, ultimately advancing our ability to combat diseases affecting geese.
V. Therapeutic and Diagnostic Implications
The comprehensive understanding derived from decoding the structure and biological role of Goose MCP carries significant practical implications for both therapeutic interventions and diagnostic advancements in goose farming and avian veterinary medicine. Targeting this pivotal viral protein offers promising avenues for disease control and prevention.
A. Vaccine Development: A Shield Against Infection
The Goose MCP is an ideal target for vaccine development, primarily because it is the most abundant and surface-exposed protein of the virion, making it the primary antigen recognized by the host's immune system. * Inactivated Vaccines: Traditionally, inactivated whole-virus vaccines have been used against GPV. These vaccines typically contain chemically or physically inactivated virions, presenting the Goose MCP in its native, antigenic conformation to the immune system, eliciting a protective antibody response. The challenge is ensuring complete inactivation while preserving antigenicity. * Subunit Vaccines: Recombinant Goose MCP, produced using molecular biology techniques, can be used as a subunit vaccine. These vaccines contain only the antigenic portions of the MCP, reducing the risk of adverse reactions and simplifying production. Identifying the key neutralizing epitopes on the Goose MCP is critical for designing effective subunit vaccines that elicit strong, protective antibody responses. This approach can also involve displaying MCP fragments on viral vectors or nanoparticles to enhance immunogenicity. * Live Attenuated Vaccines: While more complex to develop, live attenuated GPV vaccines, where the virus has been genetically modified or serially passaged to reduce its virulence while retaining immunogenicity, can offer robust, long-lasting immunity. Mutations affecting the MCP's interaction with host factors or its assembly can contribute to attenuation. The goal is to induce both humoral and cellular immunity without causing disease. * Vector-based Vaccines: The Goose MCP gene can be inserted into a non-pathogenic viral vector (e.g., fowlpox virus) or bacterial vector, which then expresses the MCP within the vaccinated bird, stimulating an immune response. This approach can offer advantages in terms of stability, delivery, and induction of both humoral and cellular immunity.
Vaccine strategies targeting Goose MCP aim to induce high titers of neutralizing antibodies that can block viral attachment and entry into host cells, thereby preventing infection and the subsequent development of Derzsy's disease.
B. Antiviral Strategies: Interrupting the Viral Cycle
Understanding the critical functions of Goose MCP opens doors for the development of specific antiviral compounds that can inhibit GPV replication. * Inhibitors of Receptor Binding: Small molecules designed to bind to the receptor-binding site on Goose MCP could prevent the virus from attaching to host cells, effectively blocking the initial step of infection. This is akin to a molecular blockade, stopping the virus before it can even enter. * Inhibitors of Capsid Assembly: Compounds that interfere with the precise protein-protein interactions required for Goose MCP subunits to assemble into a stable capsid would prevent the formation of infectious virions. Such inhibitors could target the interfaces between MCP subunits or disrupt the conformational changes necessary for assembly. * Inhibitors of Genome Packaging: Drugs that interfere with the interaction between Goose MCP and the viral genome, or with the packaging machinery, could prevent the encapsulation of genetic material, leading to the production of empty, non-infectious capsids. * Inhibitors of Uncoating: Therapeutics targeting the acid-induced conformational changes in Goose MCP that are necessary for genome release from endosomes could trap the virus within the endocytic pathway, preventing it from reaching the nucleus and replicating.
The development of such specific antivirals would offer a direct means to treat infected geese, particularly valuable in situations where vaccination is not feasible or in conjunction with vaccination to manage outbreaks.
C. Diagnostic Tools: Early and Accurate Detection
Goose MCP serves as an excellent antigen for the development of highly sensitive and specific diagnostic assays for GPV infection. * Antigen Detection ELISA: Using antibodies raised against purified Goose MCP, ELISAs can rapidly detect the presence of viral antigen in tissue samples, fecal samples, or cloacal swabs from potentially infected geese. Early detection is crucial for implementing control measures and preventing further spread. * Antibody Detection ELISA (Serodiagnosis): Purified recombinant Goose MCP can be coated onto ELISA plates to detect and quantify specific antibodies (IgY) against GPV in goose serum. This allows for monitoring flock immunity after vaccination, identifying previous exposure, and determining the prevalence of infection in a population. * Immunohistochemistry and Immunofluorescence: Antibodies against Goose MCP can be used to visualize viral antigen directly within infected tissues, providing pathological insights and confirming the presence of the virus in lesion-bearing organs. This is particularly useful for post-mortem diagnostics. * PCR-based Diagnostics: While not directly targeting the protein, the genetic sequence encoding Goose MCP is often used as a target for PCR (Polymerase Chain Reaction) assays, which detect the viral DNA itself. PCR is highly sensitive and specific for early detection of infection, even before antibody responses are detectable. Combining PCR with antibody-based detection provides a comprehensive diagnostic approach.
Accurate and timely diagnosis, facilitated by Goose MCP-based assays, is essential for effective disease management, allowing farmers and veterinarians to make informed decisions regarding biosecurity, culling, and vaccination strategies.
D. Biotechnological Applications: Harnessing Viral Proteins
Beyond disease control, the unique structural properties of Goose MCP and other viral capsid proteins present exciting opportunities in biotechnology. * Gene Delivery Vectors: Empty viral capsids, or virus-like particles (VLPs) composed solely of Goose MCP, can be engineered to encapsulate and deliver therapeutic genes or RNA molecules into specific target cells. The intrinsic ability of the MCP to bind to and enter cells can be exploited for targeted gene therapy, potentially offering a new class of delivery vehicles for avian gene editing or therapeutic protein expression. * Nanocarriers for Drug Delivery: The stable and symmetrical structure of Goose MCP-based VLPs makes them attractive candidates for nanocarocarriers, capable of encapsulating and delivering small molecule drugs, imaging agents, or other payloads with high precision. Their natural biocompatibility and potential for surface modification allow for targeting specific cell types. * Display Platforms: The surface of Goose MCP, particularly its variable loops, can be engineered to display foreign peptides or proteins. This can be utilized for developing multivalent vaccines (displaying antigens from multiple pathogens), diagnostic platforms (displaying capture agents), or even for bioconjugation in material science applications.
The therapeutic and diagnostic implications of understanding Goose MCP are profound, ranging from direct disease prevention and treatment to innovative biotechnological applications. Continued research in this area promises to yield even more sophisticated tools and strategies for safeguarding goose health and leveraging viral protein biology for broader scientific advancements.
VI. Bridging the Gap: MCP as a "Model Context Protocol"
While the primary focus of this extensive article has been on the biological "Goose MCP" – the major capsid protein of viruses affecting geese – it is important to briefly address the other significant interpretation of the "MCP" acronym: "Model Context Protocol." This concept exists in a completely different domain, specifically within the realm of computer science, artificial intelligence, and software engineering. A Model Context Protocol defines the standardized ways in which computational models, especially complex AI models, receive inputs, process information, manage their internal state, and provide outputs. It essentially outlines the "contract" for how a model interacts with its surrounding environment, including data pipelines, other models, and external applications. Such a protocol ensures interoperability, consistency, and efficient management of models within larger, distributed systems.
In today's rapidly evolving technological landscape, where AI models are increasingly integrated into diverse applications and services, the need for robust "Model Context Protocols" is paramount. Imagine a scenario where an enterprise utilizes multiple AI models for various tasks – natural language processing, image recognition, predictive analytics – each potentially developed using different frameworks and exposing different interfaces. Without a unifying protocol, integrating these models, ensuring data consistency, and managing their lifecycle becomes an incredibly complex and error-prone endeavor. A well-defined Model Context Protocol addresses this challenge by standardizing aspects such as request/response formats, authentication mechanisms, versioning, and error handling, allowing developers to interact with disparate models through a consistent interface.
This is precisely where platforms like APIPark come into play, embodying and enabling the principles of an effective "Model Context Protocol" in practical application. APIPark is an open-source AI gateway and API management platform that acts as a sophisticated intermediary, abstracting away the inherent complexities of integrating and managing diverse AI models and REST services. It provides a unified API format for AI invocation, which is a direct implementation of a Model Context Protocol. By standardizing the request data format across over 100 integrated AI models, APIPark ensures that developers and applications can interact with any AI service through a consistent interface, regardless of the underlying model's specific requirements or the platform it runs on. This standardization is crucial because it means that changes in AI models, their versions, or even prompt engineering, do not necessitate modifications in the calling application or microservices, significantly simplifying AI usage and drastically reducing maintenance costs.
APIPark further enhances this "Model Context Protocol" by allowing users to encapsulate custom prompts with AI models to quickly create new, specialized APIs, such as for sentiment analysis or advanced data processing. This feature enables developers to define unique "contexts" for their AI interactions and expose them as standardized REST APIs, ensuring that these specialized services adhere to a consistent protocol. Furthermore, APIPark offers end-to-end API lifecycle management, assisting with the design, publication, invocation, and decommissioning of APIs. This comprehensive management ensures that even when new models are introduced or existing ones are updated, the overarching protocol for interaction remains consistent and well-governed. In essence, APIPark serves as a robust framework that implements and facilitates the very notion of a "Model Context Protocol," ensuring efficiency, scalability, and ease of use in complex AI and API ecosystems for developers and enterprises globally.
VII. Future Directions and Unanswered Questions
Despite significant progress in understanding Goose MCP and viral major capsid proteins in general, numerous exciting future directions and fundamental unanswered questions remain. These areas represent fertile ground for continued research and promise to yield even deeper insights into viral biology, host-pathogen interactions, and innovative disease control strategies.
One critical area for future research is the high-resolution structural determination of Goose MCP from various GPV strains and other goose-specific viruses. While generalized parvovirus structures provide a good starting point, atomic-level structures of GPV MCP from different geographical isolates and attenuated strains would reveal subtle variations in key functional domains. This comparative structural biology could pinpoint specific residues or regions responsible for differences in virulence, host tropism (e.g., between geese and Muscovy ducks), or antigenic profiles. Such detailed structural maps are essential for rational, structure-based vaccine design and the development of highly specific antiviral compounds. Advances in cryo-EM technology, particularly in achieving higher resolutions and faster throughput, will be instrumental in this endeavor.
Another pressing question revolves around the precise identification and characterization of the host cell receptors for GPV Goose MCP. While general candidates like transferrin receptor-like molecules are often implicated for parvoviruses, definitive identification of the primary and any co-receptors in geese and Muscovy ducks is crucial. Understanding the exact molecular handshake between Goose MCP and its host receptor would unlock deeper insights into the mechanisms of host specificity and viral entry. This knowledge could lead to novel antiviral strategies that block the initial binding event, akin to a "molecular shield" preventing infection. Techniques like CRISPR-Cas9-mediated gene knockout, receptor knockdown, and affinity purification coupled with mass spectrometry will be vital in elucidating these specific interactions.
The dynamic interplay between Goose MCP and the host immune system also warrants extensive future investigation. While it's known to be antigenic, the precise mechanisms by which Goose MCP might evade or modulate the goose innate and adaptive immune responses are largely unexplored. Does it interfere with interferon signaling pathways? Does it possess domains that bind to and neutralize specific immune factors? Understanding these evasion tactics is paramount for designing vaccines that elicit superior and more durable protective immunity. Research into the specific B-cell and T-cell epitopes on Goose MCP that induce protective responses, and how these might vary between different GPV strains, will be essential for developing broadly protective vaccines against evolving viral threats. The application of advanced immunogenomic tools and single-cell sequencing techniques in geese will undoubtedly shed light on these complex interactions.
Furthermore, the non-structural roles of Goose MCP beyond its primary function in capsid formation and protection deserve more attention. Are there specific interactions between Goose MCP and host cellular proteins that influence viral replication efficiency, nuclear import, or egress? Does it play a regulatory role in viral gene expression or host cell processes? Such "moonlighting" functions, if identified, could reveal unexpected vulnerabilities for therapeutic targeting. Techniques like proximity labeling (e.g., BioID, APEX) coupled with mass spectrometry could identify novel host-protein interactors of Goose MCP in infected cells, providing clues to these broader functional roles.
Finally, the evolutionary dynamics of Goose MCP, particularly in the face of selective pressures from host immunity and vaccination programs, represent a critical area for ongoing surveillance. How rapidly does the Goose MCP sequence evolve? Are there hotspots of mutation that correlate with changes in virulence or vaccine escape? Continuous genomic sequencing of GPV isolates from outbreaks globally, coupled with detailed phylogenetic analysis of the Goose MCP gene, will be necessary to monitor these evolutionary trends. This proactive surveillance is essential for anticipating emerging strains, updating vaccine formulations, and ensuring the long-term effectiveness of disease control strategies. The convergence of structural biology, molecular virology, immunology, and bioinformatics will be pivotal in addressing these complex questions and continuing to decode the intricate world of Goose MCP.
Conclusion
The journey to decode Goose MCP has unveiled a protein of remarkable complexity and profound biological significance. As the primary building block of viral capsids affecting geese, such as the infamous Goose Parvovirus (GPV), Goose MCP stands at the nexus of viral architecture, host interaction, and immune evasion. Our detailed exploration has spanned its hierarchical structure, from the defining amino acid sequence to its intricate quaternary assembly, emphasizing the conserved beta-barrel motif and the critical role of surface loops in specificity and antigenicity. We have meticulously delved into its multifaceted biological roles, highlighting its indispensable functions in genome protection, precise host cell attachment and entry, orchestrated capsid assembly, cunning immune evasion, and its direct contribution to the pathogenesis of diseases like Derzsy's disease.
The methodologies employed to unravel these secrets are as diverse as the protein's functions, encompassing cutting-edge molecular biology, high-resolution structural biology, nuanced biophysical analyses, precise immunological assays, and sophisticated computational approaches. Each technique contributes a vital piece to the puzzle, collectively painting a comprehensive picture of Goose MCP's intricate operations. Moreover, the practical implications of this knowledge are immense, directly informing the development of more effective vaccines, guiding the search for novel antiviral therapies, and enhancing diagnostic capabilities crucial for the health and economic viability of the goose farming industry. We also briefly acknowledged the distinct meaning of "MCP" as a "Model Context Protocol" in the technological domain, highlighting how platforms like APIPark exemplify the principles of such protocols in managing complex AI and API ecosystems.
Despite the considerable strides made, the scientific journey continues. Future research promises to delve deeper into the atomic intricacies of Goose MCP from diverse strains, to unequivocally identify its specific host cell receptors, and to unravel the subtle mechanisms by which it navigates and manipulates the goose immune system. Understanding the non-structural roles of Goose MCP and monitoring its evolutionary dynamics will be equally crucial. As we look ahead, the continued investment in decoding Goose MCP and similar viral proteins will not only safeguard avian health but also enrich our fundamental understanding of virology, contributing to broader efforts in infectious disease control and innovative biotechnological applications. The Goose MCP, in its elegance and complexity, remains a captivating subject at the forefront of scientific discovery.
Frequently Asked Questions (FAQs)
1. What exactly is Goose MCP, and what is its primary function? Goose MCP, or Major Capsid Protein, primarily refers to the structural protein that forms the protective outer shell (capsid) of viruses affecting geese, such as Goose Parvovirus (GPV). Its primary function is to encapsulate and protect the viral genetic material (DNA or RNA) from degradation in the extracellular environment, ensuring the genome's safe delivery to a new host cell. Beyond protection, it also dictates host cell recognition, attachment, and entry, making it crucial for initiating infection.
2. How does Goose MCP contribute to the disease caused by Goose Parvovirus (GPV)? Goose MCP is central to GPV pathogenesis. It enables the virus to specifically bind to and enter goose and Muscovy duckling cells, dictating the virus's host tropism. Once inside, it orchestrates the assembly of new virions. The efficiency of the MCP in these processes, coupled with its role in potentially evading the host immune system, directly contributes to the severity and spread of diseases like Derzsy's disease, leading to cell damage, inflammation, and clinical symptoms in infected birds.
3. What are the key structural features of Goose MCP, and why are they important? Goose MCP exhibits a hierarchical structure. Its primary sequence of amino acids determines its identity and potential modifications. Its secondary structure (alpha-helices, beta-sheets, particularly the conserved beta-barrel motif) provides stability. The tertiary structure describes its 3D fold, forming functional domains for receptor binding, DNA interaction, and inter-subunit contacts. Finally, its quaternary structure involves the assembly of multiple MCP subunits into a stable, often icosahedral, capsid. These features are critical because they dictate the protein's ability to protect the genome, interact with host cells, and assemble into infectious virions.
4. How is the study of Goose MCP relevant to vaccine development for geese? Goose MCP is a primary target for vaccine development because it is the most abundant and surface-exposed protein of the virus, making it the main antigen recognized by the goose immune system. Vaccines (e.g., inactivated, subunit, or live attenuated) are designed to present the Goose MCP to the host, stimulating the production of neutralizing antibodies that can block viral attachment and entry, thereby preventing infection and protecting geese from diseases like Derzsy's disease. Understanding its structure and antigenicity is crucial for designing effective and broadly protective vaccines.
5. Is "MCP" always referring to a biological protein? What about "Model Context Protocol"? No, while this article focuses on the biological "Goose MCP" (Major Capsid Protein), the acronym "MCP" can also stand for "Model Context Protocol" in the realm of computer science and artificial intelligence. A Model Context Protocol defines standardized rules for how computational models interact with their environment and data, ensuring interoperability and efficient management in complex software systems. For instance, platforms like APIPark implement these principles by providing a unified API format for AI invocation, streamlining the integration and management of diverse AI models.
🚀You can securely and efficiently call the OpenAI API on APIPark in just two steps:
Step 1: Deploy the APIPark AI gateway in 5 minutes.
APIPark is developed based on Golang, offering strong product performance and low development and maintenance costs. You can deploy APIPark with a single command line.
curl -sSO https://download.apipark.com/install/quick-start.sh; bash quick-start.sh
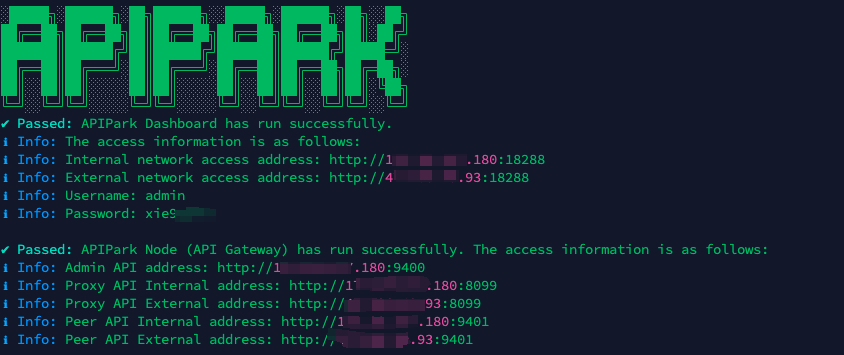
In my experience, you can see the successful deployment interface within 5 to 10 minutes. Then, you can log in to APIPark using your account.
Step 2: Call the OpenAI API.